1. Introduction
Titanium-based alloy implants have been widely used for various types of bone-anchored reconstructions due to their excellent mechanical properties and existing of the several nanometer thick surface oxide film that featured with a successful biocompatibility and corrosion resistance [1]. This naturally formed oxide layer is biologically inert, which is not conducive to bonding at bone-implant interface [2]. Vertically aligned anodic nanotubes (NT) have attracted much attention for their biofunctions, as for the improvement of osteointegration by acceleration of mesenchymal stem cells (MSCs) osteogenic differentiation [[3], [4], [5], [6]]. Moreover, the remarkable antibacterial activity of NT was also documented [7,8]. The in vivo animal models including mini pigs, rabbits and rats have all well demonstrated that NT has positively biofunctional abilities [[9], [10], [11]]. However, the fact is that implants serving used in human body is different from that in the animals as they need higher biomechanical stability due to the significant extended service in vivo. In fact, the weak bonding strength of nanotubes to the substrate has been documented, i.e. the nanotubes layer can debonding easily from titanium substrate upon encountering stress [12,13]. The debonding of nanotubes will generate debris which could lead to integration failure at the bone-implant interface. Furthermore, clinical retrieval reports mentioned that wear debris can cause mild and severe tissue reactions [14,15]. In typical case, the micro-movement between nanotube layer and the substrate when the implant is inserted into the human body, i.e. fretting will happen. This plays a vital role for inducing the failure of bone-implant interface. Wear particles generated by fretting lead to inflammation and bone resorption, and prevent osteointegration [16]. Apart from fretting wear, fretting-induced corrosion phenomenon is also another dominant factor for implants degradation. Impressively, NT decorated Ti implant is featured with improved anti-corrosion behavior than bare Ti surface in Hank’s solution and phosphate buffered saline (PBS). The improved corrosion resistance originates the thicker barrier layer of nanotubes against corrosive medium [17,18]. Clinically, titanium implants are commonly surrounded by blood-rich tissue, in which the serum proteins in the blood possibly affects implants corrosion behavior. Correspondingly, synergetic contribution from fretting wear and fretting corrosion that occurs at the bone-implant interface is an irreversible process which finally induces failure of the bone implant [19,20].
The clinical orthopaedic and dental devices are mostly designed with sharp curved surfaces, such as screw thread. These diverse surfaces cause a more complicated stress distribution on implants surface. Retrieved bone screw examinations revealed server worn thread as a result of fretting which, finally induces the mechanical failure of the bone screw. To improve the fretting resistance, we have developed a novel structure optimised TiO2 nanotubes (SONT) layer on bone screw surface by creating an interfacial layer between TiO2 nanotube and the substrate [21]. The fretting test was performed by using a self-built test platform to evaluate the anti-wear behavior of resulted SONT, compared with that of pristine bone screw and conventionally NT modified bone screws. In addition, the biocorrosion behavior of the NT and SONT decorated bone screws were also evaluated by monitoring the open circuit potential (OCP) value in simulated body fluid (SBF). The experiment results indicate that screws decorated with both NT and SONT layer exhibited reduced wear and improved anti-corrosion behavior when compared with a pristine machined bone screw (MA). Meanwhile, screw decorated with SONT layer showed more excellent wear and corrosion-resistance behavior than that of screw decorated with NT layer. It is postulated that the improvement of fretting corrosion resistance of SONT layer could be attributed to the presence of interface bonding layer, for which provide an effective isolation from corrosive environment with the substrate. To our knowledge, this work provides the first insight on fretting corrosion behavior of NT and SONT on alloyed orthopaedic screws countering with bone.
2. Materials and methods
2.1. Generate conventional and structure optimised TiO2 nanotube on screws
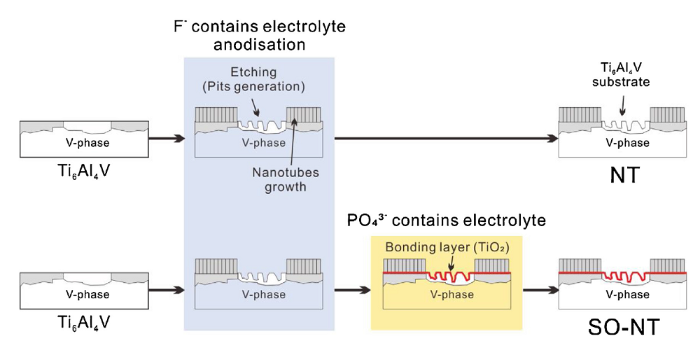
Ti6Al4V medical grade (TC4) veterinary fixation screws (3.5 mm major diameter and 26 mm length, self-tapping screw, Shanghai Huifu manufacturing Co., Ltd) were used in this study for generating NT and SONT modified bone screws. For surface modification, the screws were sequentially cleaned in ultrasonic cleaner for each 15 min with acetone (99.5 %, Fisher Scientific), ethanol (99.8 %, Fisher Scientific), and deionized (DI) water, then were dried in air. The anodization was first performed in ethylene glycol (EG, 99.5 %, ACROS Organics) with 0.5 wt% NH4F (98 %+, ACROS Organics) for 30 min under 60 V voltage, to obtain conventional NT modified screws. Followed by the first anodization, the screws were further anodized in EG solution with 3 wt% H3PO4 (85 %+, Fisher Chemical) for 1 min under 60 V to obtain SONT. The screws were then rinsed with DI water and dried with N2 stream. NT fabrication follows the same procedure as that of SONT but without additional anodization step.
2.2. Fretting test
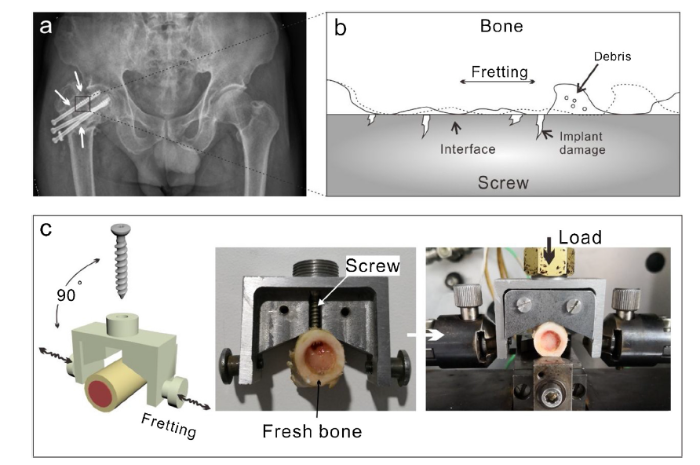
To evaluate the wear behavior of screws under fretting, a test rig setup was configured to simulate the shearing situation at bone implant interface upon applying the load vertically on screws (see Fig. 1). As shown in Fig. 1a load FN is applied on bone screws. During the fretting, debris generated and trapped at bone implant interface is illustrated in Fig. 1b and c. As the micromechanical properties are altered in dehydrated bone, in this study fresh sheep femoral bones were used as our ex-vivo fretting counter for screws [22]. The middle part of the femoral bone was collected from local abattoir within 2 h of sacrifice. The screw was inserted into the new fresh bone to form a tight contact between bone and screw interface. Before insertion, a hole with diameter of 3 mm was drilled in the center of the bone, and each self-tapping screw was inserted into each femoral bone individually. Fretting motion was examined by SRV-IV oscillating reciprocating friction and wear tester (Nye Lubricants Company). A load of 100 N was applied on screw head with 300 μm fretting distance at 37 °C. Fretting was conducted at frequency of 10 Hz and 36 × 1000 cycles test was performed.
Fig. 1.
Fig. 1.
Schematic of fretting at bone-implant interface: Fretting behavior of inserted injury fixation screws (a), load vertically applied on screws, fretting behavior at bone implant interface, the fretting leads to implant surface damage and wear debris generation (b) and mechanical jig setup (c).
2.3. Biocorrosion characterization
To investigate biocorrosion behavior, an electrochemical workstation CHI660D (CH Instruments, Inc, Shanghai, China) was used to measure the change of open circuit potential (OCP) while real-time signals of E versus time was achieved in simulated body fluid solution (SBF). The biocorrosion test consisted of an electrotank with three-electrodes (a saturated calomel electrode for reference, and a platinum foil as counter electrode), with adopting an OCP of ±250 mV and a scan rate of 0.3 mV s-1. All measurements were performed under room temperature (22 ± 1 °C). The OCP curve was recorded after the sample was soaked into SBF until stabilization (>60 min). Before the fretting test, the OCP signal was recorded for 1200 s. The comparative analysis was carried out by recording OCP value after 36 × 1000 cycles fretting test. Energy dispersive X-ray spectroscopy (EDX) was used to analyze the chemical composition of samples at different areas on specimens.
2.4. Fretting track observation
2.4.1. Optical examinations
Optical observation is an intuitive and widely applied method to evaluate severe wear or corrosion for orthopaedic retrieval implants analysis. As the dark grey color of nanotubes layer is significantly different with titanium alloy burnished surface, optical investigations can be a useful tool to investigate the general fretting of the three samples.
2.4.2. Wear observation at tapping areas
According to shear stress generated during insertion, which concentrates at the thread ridge areas of the screw, where the most damaged occurred during insertion by countering the bone. The repetitive shear stress can weaken the bonding of the TiO2 nanotube layer and lead to TiO2 nanotubes debonding from the screw substrate. Therefore, the tapping areas of three samples were examined by SEM, and the nanotubes integrity on NT and SONT screws were investigated and compared. A FESEM (JEOL-7601 F, JEOL, Japan) was operated at voltage of 5 kV for topography observation of screws before and after fretting test. The topography of MA, NT and SONT were examined and compared under the same magnification. Under the selected tapping areas, nanotubes damage on NT and SONT could be compared obviously.
2.4.3. NT and SONT integrity on the thread ridges
Ascribed to the scale of nanotubes, conventional wear quantitative methods, for example weight loss is not a suitable method to measure the fretting wear of the nanotubes. Furthermore, wear damage of the screw surface could widely occurrs on different locations of whole screw. Herein, a methodology was proposed to quantify nanotubes damage on NT and SONT screws.
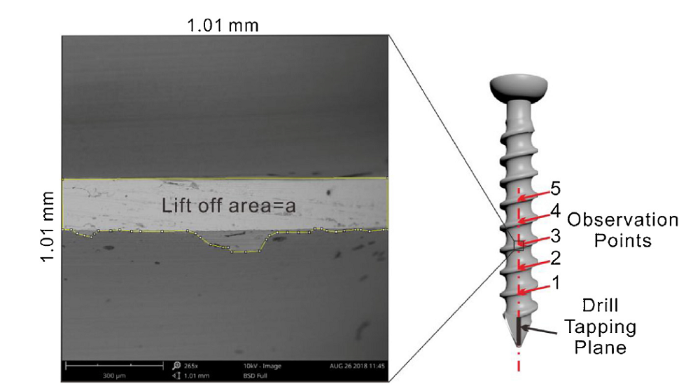
Fig. 2 illustrates the locations observed by SEM on the self-tapped screw. The damage of nanotubes was mainly concentrated on thread ridge at points 1, 2, 3, 4 and 5 on the thread. The five points were defined as the areas passed through the central line of the screw (the line passing along the drill tapping plane) and the proportion of debonding areas were measured by Image J software 1.47 V (National Institutes of Health). Under SEM scanning area in every location (1.01 × 10.1 mm2), the actual nanotubes debonded areas (see Fig. 2) were calculated. With the average actual peeling off areas value of five points, one circle of thread lift off area (φ) can be calculated by Eq. (1), based on the hypothesis that the wear debonding areas was equal on different regions on thread.
Where, φ is the lift off area on one circle of thread, mm2; ai (i = 1, 2, 3, 4, 5) is each observation points of actual nanotubes lift off area, mm2; r is the major radius of thread with 1.75 mm. Statistical analysis of volume loss was performed by one-way ANOVA.
Fig. 2.
Fig. 2.
Schematic of self-tapping screw and SEM observation of debonding of TiO2 nanotube layer, five points on the thread ridge were used for SEM examinations.
However, the method above described is unable used to work out the debonding area of MA sample as no TiO2 nanotube layer on the MA screw, as such the damage areas could not be determined by SEM observation. Therefore, the fretting of titanium screw (MA sample) was only topographically compared with NT and SONT screws.
2.4.4. MA, NT and SONT thread width measurement
As insertion procedure generated stress and harsh wear at thread ridge, the tips were damaged more than other locations, the material on thread tip would be mechanically removed. Therefore, worn threads become thicker than pristine thread, MA, NT and SONT thread width were measured by SEM (JEOL-7601 F, JEOL, Japan) and ImageJ 1. 47 V (National Institutes of Health) after the fretting test.
3. Results
3.1. TiO2 nanotube layer
NT and SONT exhibited a optical homogeneous dark grey color without obvious defects. The micro topography of MA, NT and SONT are shown in Fig. 3. It was observed from the SEM examination that the machined groove track displayed a rough topography under micro scale on MA screw. In contrast, there were non-machining grooves on both NT and SONT specimens. However, vertically arranged nanotubes structure on NT and SONT were observed. Micro/submicro sized pits were formed on both NT and SONT layers.
Fig. 3.
Fig. 3.
FESEM images of the surfaces of MA, NT and SONT screws. The micro pits were formed on both NT and SONT surface (black arrows).
Fig. 4 shows the high resolution of SEM examinations of both NT and SONT screws. It was observed that the nanotube diameters of both NT and SONT were 100 nm in average, with a length (nanotubes layer thickness) of 2.5 ± 1 μm. SONT exhibited a 120-150 nm thick bonding layer at nanotubes bottom, whereas NT exhibits a typical nanotubes spherical bottom. The SEM examinations revealed the shape of pits varies, and distributed randomly within both NT and SONT layers, the details of pits are shown in Fig. 4a1.
Fig. 4.
Fig. 4.
FESEM images of NT and SONT, top view of NT (a1) and SONT (b1), side view of NT (a2) and SONT (b2) and the bottom part view NT (a3) and SONT (b3). The structure of NT and SONT were shown highly similarity except the bottom, a flat layer was bonded nanotubes bottom in SONT.
Meanwhile, the chemical compositions of both NT and SONT at the pit areas were examined by EDX and were shown in Fig. 5. It was observed that the chemical composition of nanotubes of both NT and SONT exhibited 56-57 % of oxygen and 23-24 % of titanium, indicates TiO2 composition. However, the NT pits areas presented 54 % titanium, 21 % fluorine and no oxygen was detected. While as the elements on SONT pits areas showed high similarity with that on nanotubes areas. Based on results, it is speculated that chemical composition of pits on NT was titanium substrate, and fluorine concentrated at the pits area.
Fig. 5.
Fig. 5.
SEM-EDX images of NT and SONT at pits and nanotubes areas: NT pits areas and nanotubes areas (a) and SONT pits areas and nanotubes areas (b).
3.2. Corrosion behavior of MA, NT and SONT modified screws
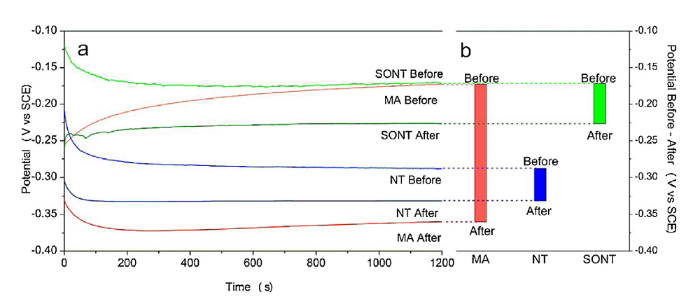
Before fretting, the OCP of both SONT and MA screws were stabilized at - 0.17 V vs. SCE, exhibited low tendency to corrosion (Fig. 6a). By contrast, NT presented the lowest OCP value of - 0.28 V vs. SCE. These results are in contrary with the previous studies of anodization on pure titanium [17]. The reason responsible for this can be attributed to that the anodization in NH4F undermined the pristine machinery titanium surface, leading to a lower OCP value. Impressively, as the presence of bonding layer, SONT proved highest corrosive resistance, as indicated OCP value.
Fig. 6.
Fig. 6.
Open circuit potential of MA, NT and SONT: before and after fretting (a) and OCP decreased value of each screws after fretting (b).
After fretting test, the worn screws were immersed into fresh SBF. Both NT and SONT screws displayed decreased OCP values. Interestingly, remarkable decrease of the OCP value was observed for MA screw. As the natural titanium oxide is mechanically depassivated during fretting (or removal during insertion), the titanium substrate of MA screw sufficiently exposed into corrosive environment, leading to decrease of OCP value from -0.17 V to -0.36 V vs. SCE (see Fig. 6b). In contrast, the decreased OCP value of NT was only approximately 0.04 V vs. SCE, from -0.29 V to -0.33 V vs. SCE. Meanwhile, the OCP value of SONT screw decreased around the same value as 0.05 V vs. SCE from -0.17 V to -0.22 V vs. SCE. However, SONT sample exhibited the highest value of OCP even after fretting, indicating the mechanical stability of SONT bonding layer to protect titanium against corrosion.
3.3. Fretting track observation
3.3.1. Optical general investigation
Optical images demonstrated that the most of nanotube film on both NT and SONT screws were maintained and no severe damage was observed during the fretting test, as revealed in Fig. 7. Meanwhile, obvious nanotubes debonding to expose the alloy substrate were observed mainly at thread ridge. There was no obvious worn marks on the thread ridge bottom. Obvious substrate exposure was also detected at the tapping areas on both NT and SONT screws as shown in Fig. 7b2 and b3, respectively. Moreover, nanotubes on NT threads ridge have deboned, whereas the nanotubes on SONT were less damaged.
Fig. 7.
Fig. 7.
Optical image of MA, NT and SONT screws before fretting test (a1) and after fretting test (a2). The enlarged optical images revealed the fretting worn markers on pristine (b1), NT (b2) and SONT (b3) screws, respectively.
3.3.2. Fretting wear evaluation at tapping areas
Fig. 8 illustrated the worn marks of the drill tapping areas on three screws worn, suggesting the tapping areas may have been possibly damaged during the initial insertion. Figures 8b1 and 8c1 showed that patches of the nanotubes on NT and SONT screw tapping areas were damaged. In contrast, Fig. 8b2 and c2 indicated that the nanotubes layer on SONT was more integrated as it remained in the same area. On tapping plane, NT screws exposed more substrate areas than that of SONT (Fig. 8b1, b2, c1 and c2, white arrow). MA surface has been worn on areas due to the presence of surface asperity. Fig. 8a1 and a2 demonstrated that the worn on MA screw was heavy and scathes were widely distributed on tapping areas. Ascribed to the presence of nanotubes, NT and SONT screws surfaces displayed a slight worn than that of MA screw.
Fig. 8.
Fig. 8.
Worn after fretting on drill tapping areas, MA screw a1 and a2, NT screw b1 and b2, SONT screw c1 and c2. MA tapping area has worn heavily, tapping upper areas were also worn (a1 and a2, white arrows). The nanotubes layer lift off on NT tapping areas were more than that on SONT (b1 and c1, b2 and c2), some areas on SONT displayed light grey color (c1 and c2, white arrows).
3.3.3. NT and SONT integrity on thread ridge
As shown in Fig. 9 (MA 1-5), the MA screw thread ridge was worn heavily. Apart from thread worn, the damage was also remarkable on areas both above and beneath the threads. However, severe wear on NT and SONT were not observed, as showed in Fig. 9. This may be attributed to the presence of protective nanotubes layer. Worn areas of NT and SONT screws were mainly distributed adjacent the thread ridge as observed in Fig. 9. It was also observed that all the nanotubes layer on five thread ridges (from NT-1 to NT-5) were removed from the substrates. Furthermore, areas near the thread tips appeared delaminated from the substrate and led to the exposure of the substrate (see Fig. 9 NT-2, NT-3 and NT-4, white arrow). It is postulated that SONT exhibited an improved mechanical stability, as confirmed by the SEM observation that the nanotubes remained even in some thread areas (SONT-2 and SONT-5, black arrow). Moreover, some areas of destroyed nanotubes displayed light gray (different with the substrate), indicating that the bonding layer still covered the titanium substrate (SONT-1 to SONT-5, red arrow).
Fig. 9.
Fig. 9.
SEM observations of worn thread ridges of machined (MA 1-5), NT (NT 1-5) and SONT (SONT 1-5) screw, bar = 300 μm.
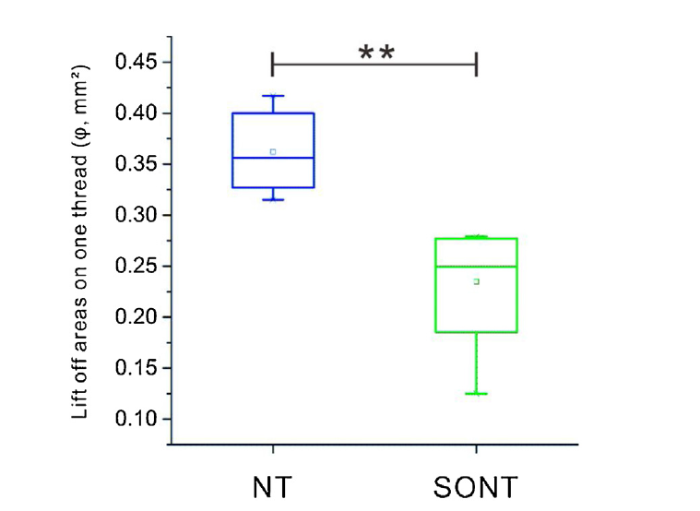
Use the methodology of nanotubes quantitative analysis illustrated in Eq. (1), the deboned areas on every thread of NT and SONT were calculated. Fig. 10 showed the debonding areas on one thread (φ) of NT and SONT screw thread tip. As the p value was 0.0051, SONT sample exhibited an average debonding areas of 0.23 mm2 while it was 0.36 mm2 for NT sample. As calculated, the debonded areas on thread of the SONT decorated screw decreased ~36.11 % compared with NT screw, indicating an improved anti-fretting wear for SONT nanotubes layer. Especially, among the five SEM selected points, it was noted that SONT-5 had a lowest worn of only 0.06 mm2.
Fig. 10.
Fig. 10.
Lift off areas of nanotubes layer on thread for NT and SONT decorated screw, **, p < 0.01. p value: 0.0051, Error bars represent standard error with the mean (s.e.m).
3.3.4. MA, NT and SONT screw thread worn width measurements
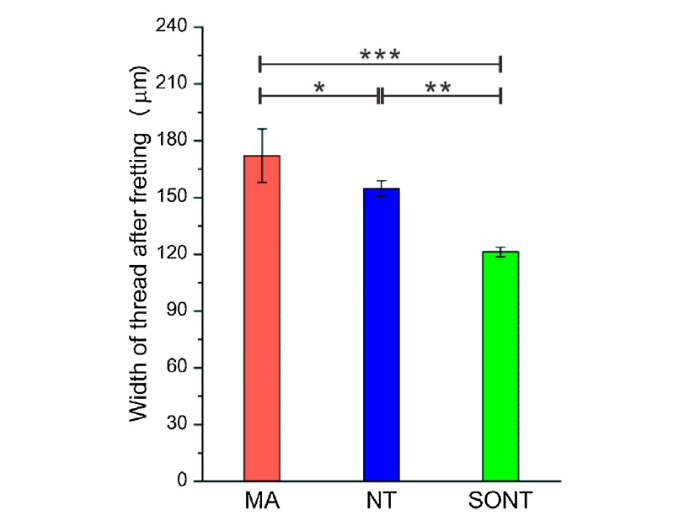
Fig. 11 illustrates the selective measurement points (marked with 1, 2 and 3) on worn thread tracks of MA, NT and SONT screws. It revealed that the MA screw showed the highest value of thread width of 172.13 μm in average, while the NT screw reached 154.81 μm and 121.25 μm for the SONT screw (Fig. 12). Static analysis shows significant difference between width of NT thread and that of MA with p = 0.036, while the p value of NT width with SONT width is 0.0072. The SONT screw thread width was minimum worn, and exhibited the narrowest thread worn track after fretting test.
Fig. 11.
Fig. 11.
Thread width measure points (white bar areas) of MA(a), NT(b) and SONT(c), bar = 300 μm.
Fig. 12.
Fig. 12.
Thread width of MA, NT and SONT after fretting, *, p < 0.05, **, p < 0.01, ***, p < 0.001.
4. Discussion
Implants with appropriate initial contact to adjacent bone generate physical stress at the interface, while the shear generates relative micromotions. The continuous micro-motion accelerated by biological corrosion (fretting corrosion) generates foreign debris, which is the main cause for chronic inflammatory and/or aseptic loosening [[23], [24], [25], [26]]. Although the NT and SONT were successfully generated on TC4 medical screws, micro pits were observed on both surfaces, which was most likely due to erosion of the vanadium oxide during the anodization in fluorine containing electrolyte [6,27]. The EDX examination well proved the presence of bonding layer at micro pits areas. On nanotubes areas, both NT and SONT contained above 55 % of oxygen, 23-25 % of titanium, 10 % fluorine, 7-8 % carbon and less than 3% vanadium, as reported in Fig. 5. However, the chemical composition in pits area showed a significant difference. For the pits on NT screw contain 54 % titanium, 21 % fluorine, 17 % carbon, 5% aluminum and 3% vanadium, indicating the composition of alloy substrate without the presence of large amount oxides. Furthermore, the fluorine content was two folds higher than that of nanotubes areas. Due to selective dissolution during anodization, the phase containing vanadium (β phase) was further enhanced with etching in fluorine containing electrolyte, due to the high solubility of vanadium oxide [27]. The chemical composition of pits on SONT contains the same amount of oxygen, titanium, fluorine in comparison with that of nanotubes areas, confirming the formation of one layer of titanium oxides at pits areas.
The lower OCP value of NT on Ti6Al4V was due to the micro pits formed during anodization. The formed pits were 'channels' linking corrosive biofluid to substrate while immersed in SBF environment, leading to NT surface corrosion. The improvement of corrosion resistance of SONT was due to existing of bonding layer at the interface of nanotubes and alloy substrate. From the initial OCP value of SONT, the bonding layer sealed the micro pits at bottom of nanotubes to obstruct the aforementioned 'channels' (Fig. 13).
Fig. 13.
The vanadium containing phase dissolved during nanotubes generation process that caused micro pits formed, leading to substrate exposure. The bonding layer presents a vital role to against corrosion in SBF though sealed the pits.
European Union via the H2020-MSCA-RISE-2016 program (BAMOS, 734156)
The presence of interfacial bonding layer not only improved corrosion resistance, but also enhanced the tribological performance of SONT layer. Orthopaedic screws can be even damaged both during the initial insertion procedure and encountering long-term fretting in vivo. As self-tapping screw’s thread faces with mass stress and heavy rubbing during drilling and installation, the cortical bone can mechanically remove the material on the thread ridge. From the contrastive SEM images of the tapping areas, SONT has the minimum debonding areas.
After insertion, due to the full mechanical contact at bone-thread interface, threads tips have a more severe fretting in some areas than others. The SONT exhibited 40 % less of nanotubes debris released than that of NT. Although the damage amount was only at micro scale (μm2) level, the detached nanotubes from substrate might form a micro sized cluster that would be released into the bone and cause tissue reactions. in vivo tissue reactions for TiO2 nanotubes are still barely studied, nevertheless the micro-sized wear debris released into tissue have been widely proven to be toxic.
Furthermore, TiO2 nanotubes can effectively cover alloy substrate, for which plays a vital role in protecting titanium implant. Meanwhile, any NT detachment can lead to direct exposure of alloy substrate into the biocorrosive liquid under in vivo atmosphere, leading to release of metal ions from the areas. Moreover, the experimental results of thread widths after fretting test of the three screws also proved that the SONT had the highest capability to protect worn thread, as debris generated could be easily trapped at interface.
5. Conclusion
The NT and SONT were generated on medical grade Ti6Al4V (TC4) screws to fabricate TiO2 nanotube-decorated bone fixation. The fretting and corrosion behavior of the modified screws were investigated by an ex-vivo bone screw fretting simulation system and OCP measurement carried out in SBF. The results demonstrated that the screw decorated with SONT exhibits great potential to be applied on orthopaedic devices. The conclusion can be drawn as follows:
(1)Before fretting test, the OCP value of NT was lower (-0.29 V) than that of Ma surface (-0.17 V), indicates that NT on alloy has more tendency to erosion. However, the present of NT film protected the titanium substrate during fretting process as OCP decreased only 0.04 V. Impressively, OCP of MA screw decreased significant after fretting (decreased 0.19 V), exhibited a poor mechanical stability of bare alloy surfaces.
(2)SONT displayed the highest OCP value both before (-0.17 V) and after fretting (-0.22 V). Impressively, the OCP value of SONT after fretting was still higher than that of the NT before fretting.
(3)TiO2 nanotubes layer has been well proved its protective function against fretting damage on NT and SONT screws, both screws ridges have significantly less worn than Ma screw.
(4)SONT exhibited significantly enhanced fretting resistance than NT. Thus, SONT displays an enhanced fretting resistance compared to NT and Ma screws.
Acknowledgments
This work is financially supported by the European Union via the H2020-MSCA-RISE-2016 program (BAMOS Project, 734156), Royal Society via the International Exchange Program (IE161349), Key Research Project from the National Key Research and Development Program of China (2016YFC1100401), National Natural Science Foundation of China (51705507), and Young Elite Scientists Sponsorship Program by CAST (2017QNRC0181).
Reference
DOI
URL
PMID
[Cited within: 3]

Titanium dioxide (TiO(2)) nanotubes can accelerate the adhesion and differentiation of osteoblasts, yet little is known how this nano-modified implant surface affects osseointegration at molecular level in vivo. The aim of this study was to investigate the effects of TiO(2) nanotubes with different diameters (30 nm, 70 nm and 100 nm) on biological attachment mechanism of implants to bone in vivo by studying the gene expression and bone formation around the implants. The histological features and fluorochrome labeling changes of bone around implants on the non-decalcified sections (at 3, 5 and 8 weeks after implantation) were investigated by using traditional light- and fluorescent microscopy, and the gene expression of alkaline phosphatase (ALP), osterix (Osx), collagen-I (Col-I) and tartrate-resistant acid phosphatase (TRAP) was examined by using real-time PCR at 1, 2, 3, 4 and 5 weeks after implantation. Comparing with machined titanium implants, a significant increase in bone-implant contact (BIC) and gene expression levels was found in the bone attached to implants with TiO(2) nanotubes, especially with 70 nm diameter nanotubes. At the same time, the sequential fluorescent labeling images illustrated dynamic bone deposition. In conclusion, TiO(2) nanotubes can modulate bone formation events at the bone-implant interface as to reach favorable molecular response and osseointegration; in addition, the diameters of nanotubes can be precisely controlled in order to obtain better bone formation.
DOI
URL
PMID
[Cited within: 3]

Titanium and its alloys are commonly used for dental implants because of their good mechanical properties and biocompatibility. The surface properties of titanium implants are key factors for rapid and stable bone tissue integration. Micro-rough surfaces are commonly prepared by grit-blasting and acid-etching. However, proteins and cells interact with implant surfaces in the nanometer range. The aim of this study was to compare the osseointegration of machined (MA), standard alumina grit-blasted and acid-etched (MICRO) and nanostructured (NANO) implants in rabbit femurs. The MICRO surface exhibited typical random cavities with an average roughness of 1.5 mum, while the NANO surface consisted of a regular array of titanium oxide nanotubes 37+/-11 nm in diameter and 160 nm thick. The MA and NANO surfaces had a similar average roughness of 0.5 mum. The three groups of implants were inserted into the femoral condyles of New Zealand White rabbits. After 4 weeks, the pull-out test gave higher values for the NANO than for the other groups. Histology corroborated a direct apposition of bone tissue on to the NANO surface. Both the bone-to-implant contact and bone growth values were higher for the NANO than for the other implant surfaces. Overall, this study shows that the nanostructured surface improved the osseointegration of titanium implants and may be an alternative to conventional grit-blasted and acid-etched surface treatments.
AbstractWe report for the first time on mechanical properties of anodically formed, self-organized TiO2 nanotube layers on titanium. We compare their behavior in their virgin state and after conversion to their semi-metallic form by an acetylene treatment. We show that the acetylene treatment leads to significant enhanced hardness, tensile strength and friction behavior.]]>
Implants are widely used for orthopaedic applications such as fixing fractures, repairing non-unions, obtaining a joint arthrodesis, total joint arthroplasty, spinal reconstruction, and soft tissue anchorage. Previously, orthopaedic implants were designed simply as mechanical devices; the biological aspects of the implant were a byproduct of stable internal/external fixation of the device to the surrounding bone or soft tissue. More recently, biologic coatings have been incorporated into orthopaedic implants in order to modulate the surrounding biological environment. This opinion article reviews current and potential future use of biologic coatings for orthopaedic implants to facilitate osseointegration and mitigate possible adverse tissue responses including the foreign body reaction and implant infection. While many of these coatings are still in the preclinical testing stage, bioengineers, material scientists and surgeons continue to explore surface coatings as a means of improving clinical outcome of patients undergoing orthopaedic surgery. (C) 2013 Elsevier Ltd.
DOI
URL
PMID
[Cited within: 4]

It is well known that the growth of osteoblast cultured on titanium with nanotube layers can be significantly increased compared to unanodized surfaces. In the current study, the corrosion behavior of titanium with nanotube layers was studied in naturally aerated Hank's solution using open circuit potentials (OCP), electrochemical impedance spectroscopy (EIS) and potentiodynamic polarization tests. The electrochemical results indicated that TiO2 nanotube layers on titanium showed a better corrosion resistance in simulated biofluid than that of smooth-Ti. The OCP, corrosion rate (I(corr)), passive current density (I(pass)) and the oxygen evolution potential (E(o)) were significantly influenced by titanium oxide nanotube layers acquired by anodization. The anatase nanotube layer showed higher OCP and smaller current density than the amorphous nanotube layer. EIS analysis showed that the annealing had a significant effect on the corrosion resistance of the outer tube layer (R(t)), but little effect on the corrosion resistance of the inter-barrier layer (R(b)) for nanotube layers. The results suggested that titanium with TiO2 nanotube layers has an adequate electrochemical behavior for use as a dental implant material.
DOI
URL
PMID
[Cited within: 3]

Titanium oxide nanotubes prepared by anodization have received considerable attention in the biomaterials domain. The objective of this study was to demonstrate the electrochemical behavior of different diameter TiO(2) nanotube layers on titanium in phosphate buffered saline (PBS) and Dulbecco's minimum essential medium+10% fetal calf serum (D-FCS) using open circuit potentials (OCP), electrical impedance spectroscopy (EIS), and a potentiodynamic polarization test. The results showed that the nanotubes had higher OCP, higher resistance of the inter barrier layer (R(b)), and lower I(pass) in the two test solutions compared to the smooth Ti, especially the 30 nm diameter nanotubes. The corrosion resistance of the nanotubes in D-FCS was higher than in PBS because of protein adsorption from the D-FCS solution as suggested by scanning electron microscope (SEM) images. In addition, protein aggregates of 30 nm diameter nanotubes caused the model of EIS spectra to transform from two-layer to three-layer. The corrosion behavior of the nanotubes for use as a dental implant material is discussed.
DOI
URL
PMID
[Cited within: 3]

The immune system modulates many key biological processes in humans. However, the exact role of the immune system in particle-associated periprosthetic osteolysis is controversial. Human tissue retrieval studies, in vivo and in vitro experiments suggest that the immune response to polymer particles is non-specific and macrophage-mediated. Lymphocytes may modulate this response. However direct lymphocyte activation by polymer particle-protein complexes seems unlikely. However, metallic byproducts may complex with serum proteins and lead to a Type IV, lymphocyte-mediated immune reaction. In predisposed individuals, this reaction may rarely lead to persistent painful joint effusions, necessitating debridement and excision of the bearing surfaces of the prosthesis. In these patients, retrieved periprosthetic tissues exhibit histological evidence of perivascular lymphocytic cuffing. These findings are worrisome, given the fact that increasing numbers of metal-on-metal joint implants are being implanted in younger more active individuals worldwide.
DOI
URL
PMID
[Cited within: 3]

The two leading causes of failure for joint arthroplasty prostheses are aseptic loosening and periprosthetic joint infection. With the number of primary and revision joint replacement surgeries on the rise, strategies to mitigate these failure modes have become increasingly important. Much of the recent work in this field has focused on the design of coatings either to prevent infection while ignoring bone mineralization or vice versa, to promote osseointegration while ignoring microbial susceptibility. However, both coating functions are required to achieve long-term success of the implant; therefore, these two modalities must be evaluated in parallel during the development of new orthopaedic coating strategies. In this review, we discuss recent progress and future directions for the design of multifunctional orthopaedic coatings that can inhibit microbial cells while still promoting osseointegration.
DOI
URL
PMID
[Cited within: 4]

The present work reports the fabrication of self-organized porous oxide-nanotube layers on the biomedical titanium alloys Ti-6Al-7Nb and Ti-6Al-4V by a simple electrochemical treatment. These two-phase alloys were anodized in 1M (NH(4))(2)SO(4) electrolytes containing 0.5 wt % of NH(4)F. The results show that under specific anodization conditions self-organized porous oxide structures can be grown on the alloy surface. SEM images revealed that the porous layers consist of arrays of single nanotubes with a diameter of 100 nm and a spacing of 150 nm. For the V-containing alloy enhanced etching of the beta phase is observed, leading to selective dissolution and an inhomogeneous pore formation. For the Nb-containing alloy an almost ideal coverage of both phases is obtained. According to XPS measurements the tubes are a mixed oxide with an almost stoichiometric oxide composition, and can be grown to thicknesses of several hundreds of nanometers. These findings represent a simple surface treatment for Ti alloys that has high potential for biomedical applications.










 WeChat
WeChat